High Quality vs Low Cost Bargain: The Current Dichotomy! (XXIII). How to compare COVID19 Vaccines prices?
Last Friday, we left you with an extensive homework to read. Some of the articles were didactical, others more specialized with the scientific or medical jargon; meanwhile the rest were quite informative and favorable for you to grasp the situation of the COVID19 vaccines. I hope you enjoyed your reading. My objective was to set all my readers and our minds in the same page. Once the pandemic started, the laboratory pharmaceutical industry started a race to find different alternatives of diagnostics, antibody medicines, therapeutics or palliative treatments and preventive vaccines. A race to develop all these options started as of March 2020, and we all have been witnesses of this race, with what it seems successful and the failures (or limited outcomes) at each of these COVID19 medical solutions.
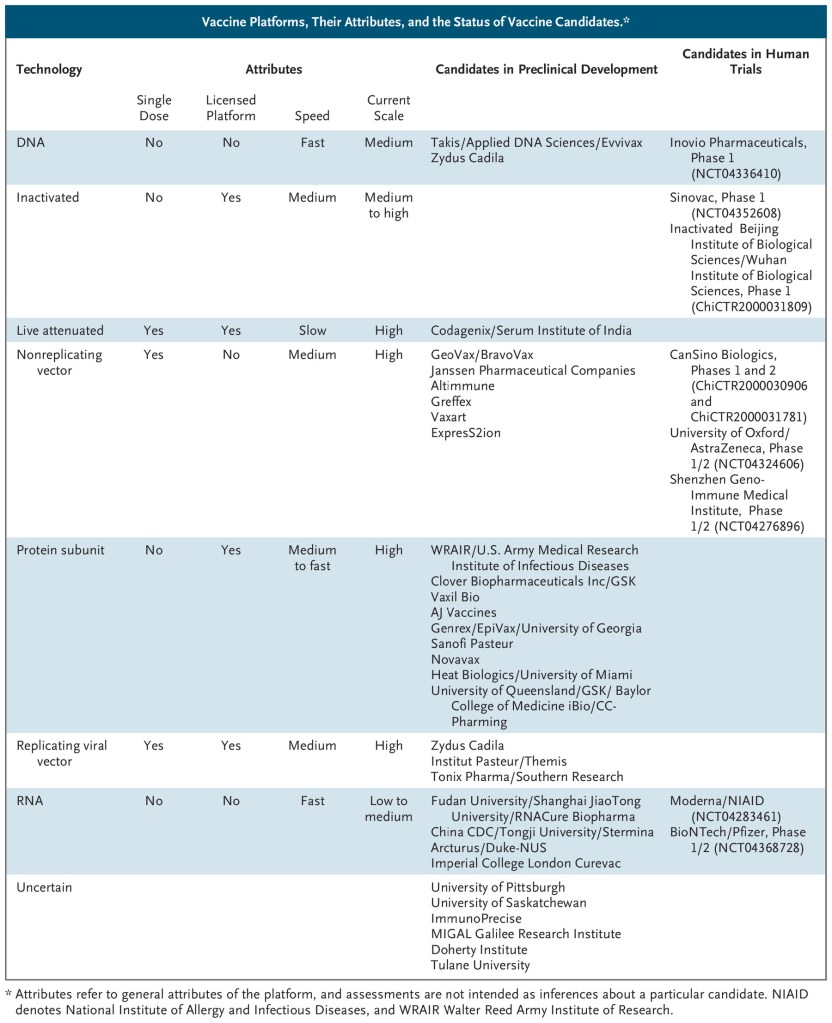
When we land in the field of prevention vaccines, these can be classified under the following categories as:
- Live Attenuated Vaccines (virus classical traditional approach)
- Inactivated Vaccines (virus classical traditional approach)
- Viral Vector Vaccines, which can be of two types, (1) Replicating; and (2) Non-replicating.
- Protein Based Vaccines, which can be of two types, (1) Protein sub-unit, and (2) Virus Like Particles (VLP).
- Genetic or Nucleic Acid Approach Vaccines: These could be (1) DNA or, (2) messenger RNA (mRNA) technologies
- Others like, Neutralizing Antibodies Vaccines, for example.
I have gathered the following general table prepared by McKinsey Consulting House. To this season, this is the status of the vaccines authorized in the first quarter of 2021 by the FDA-EUA (Emergency use authorization).

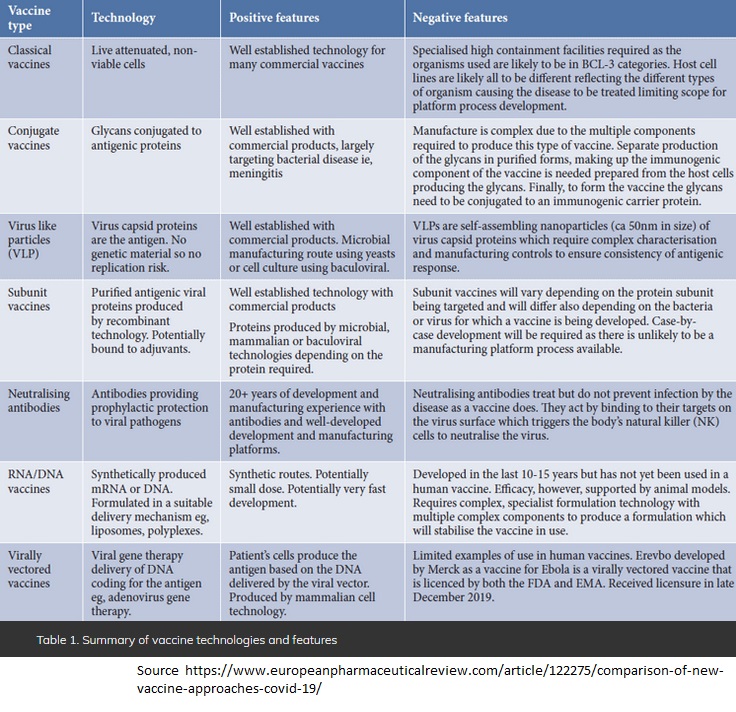
Before proceeding further, I wish to acknowledge that all these tables of comparison that I will include today, have been beautifully well researched by the authors that I have cited on the list of readings that I prepared for you last Friday. After your scanning scrutiny to all these websites shared, I am sure you already know the differences between all these approaches when it comes to understanding the release of vaccines. I won’t judge nor provide any strategic statement to the traditional classical approaches to vaccines. First, because the pharmaceutical industry is involved in their development since at least a century ago. But, there are two new approaches which are practically recently fresh in the market. So new, or extremely recent. And these are the genetic and the viral vector vaccines, which were the first ones to be released and approved under the Emergency Use Status by the FDA-EUA in December last year.
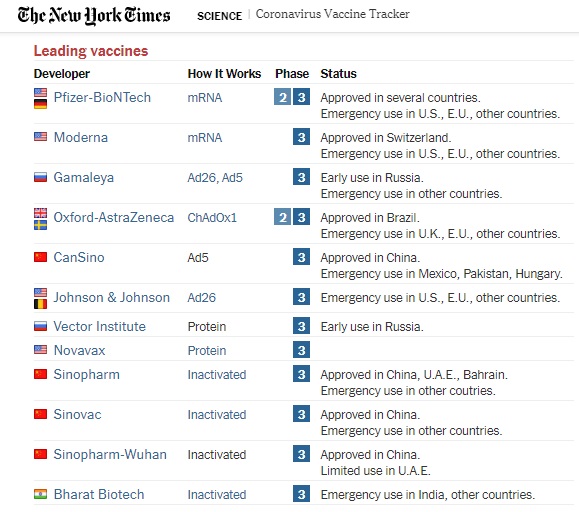
The genetic vaccines already authorized by the FDA-EUA under this emergency are the Pfizer-BioNTech (mRNA), Moderna (mRNA). And, in February 27th, the Johnson &Johnson vaccine (Viral Vector Vaccine) received the green light from the FDA-EUA. Another viral vector vaccine famously approved in the UK, is the Oxford Astra-Zeneca. To this day there are more than 270 vaccines in the process of clinical development under different phases, each running the race to arrive to the regulatory pathway for emergency authorization.

Regardless the type of vaccine, what has happened during this pandemic is that the scientists and the whole world aligned its needs and wants to the urgency of vaccinating our populations. The alignment between the emergency, with the possibilities and opportunities; coming from the giga funding and the power of strong financing under the pressure for a global cure. It has been an unprecedented challenge to be an observer of this racing to develop COVID-19 vaccines. Either we like it or not, it has happened on an exceptional and extraordinary record timeline.
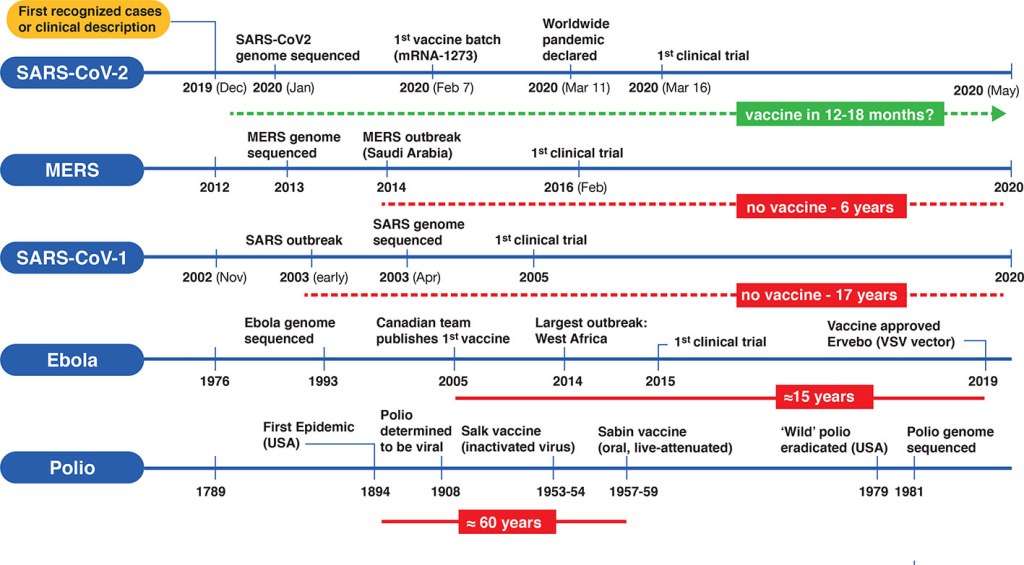
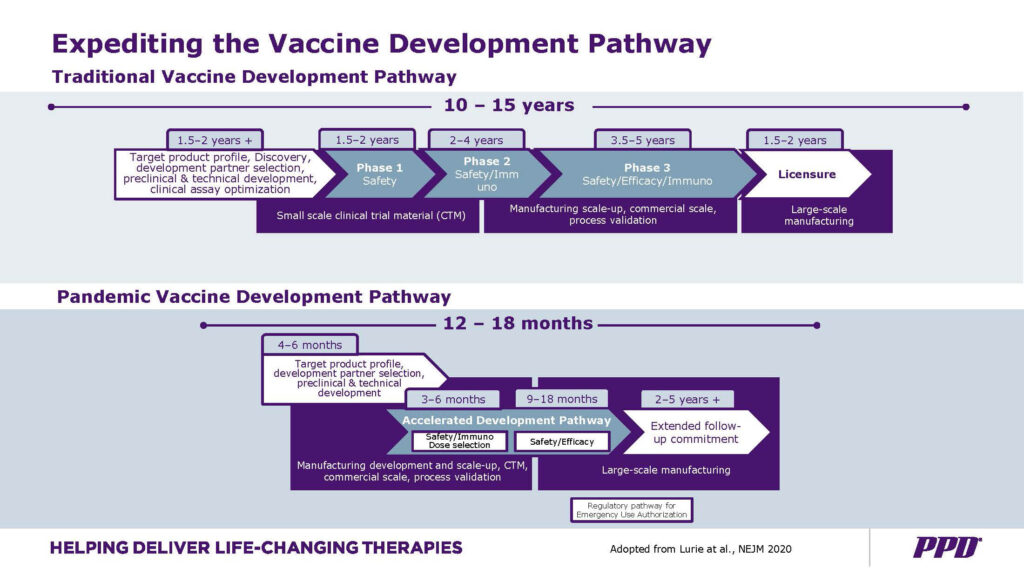
From the outside of the pharmaceutical industry, it also looks unbelievable to see how the traditional paradigm of performing a vaccine, under this specific context of the pandemic, has shorten the process of vaccine development from 10 to 15 years to less than 12 months! And what amazes me the most is: underneath everything, it seems that in the current context of balancing between the risks vs benefits of these new vaccine technologies; the genetic vaccines, were the first ones to rise and get the emergency permits from the regulatory entities. Something so new and unknown when it comes to long term effects. Regardless the unproven dangers inherent to the overlapping phases to shorten the new genetic and viral vector vaccine development time, the three top vaccines already being inoculated (Pfizer-BioNtech, Astra Zeneca and Moderna) are going elevate us from the crisis, or given their unknown long term effects, these will also affect us for the long run. Will this speedy development pace contribute to our health for the long run or not? My answer is we do not know.
How safe is to get a COVID19 vaccine under these circumstances? As I mentioned it to you previously, in relation to the United States of America, these vaccines were approved under an emergence use authorization (EUA). To this day in the United States, the emergency authorized vaccines by the FDA are simply three. Other countries have approved their vaccines under their own regulations. The risk with the new technologies that were approved and are being injected massively, is the unknown. Really, true, we do not know. For example the genetic vaccines (mRNA) have never been done before, and never in our history as human beings, in less than a year. Even though these vaccines are approved for emergency use (EUA), there are not licensed DNA or RNA existing vaccines utilized in the past. The viral vector approach method (used to develop Astra-Zeneca, CanSino Biologics and Johnson&Johnson), at least has been proven previously in veterinary medicine and with Ebola. But still there are certain risks associated with them. Notwithstanding the type of vaccines developed under other sicknesses and previous circumstances, we have to be aware that each type of vaccine has its advantages (positive features) and disadvantages (negative features). And depending on the individual profile of the patient, each of us has to open our eyes and check first, all the consequences that each of us may bear if we accept to get vaccinated with new vaccines. Particularly, those with compromised immune systems or with previous diseases such as cardiovascular issues, diabetes, children and teenagers below 18 years old, and/or reproductive people who are still planning to have babies, etc.
Pricing Contradictions in the case of COVID19 Vaccines. Now that we have analyzed the issues with the current (up to date) vaccines, I wish to begin to show you the principles of a pricing war. I will show you theoretically, with the application of the dynamic theory of pricing. I will use an example of the existing prices of COVID19 vaccines which have been approved for commercialization and distribution under an emergency use authorization. As I mentioned previously, pricing wars happen when competitors, begin clash on price only, and the cannibalization occurs when reducing the prices below the Value Equivalent Line (VEL). To this day, we can only compare prices of those vaccines that were the first movers, see the slide below:

I will leave this publication here. We will continue tomorrow. I will finish this saga on Wednesday 31st. All the bibliography of today is included in the readings provided last Friday. All the best, and wishing you a beautiful Easter time.

Any high quality mind will always elevate people and its societies, not the other way around.
Eleonora Escalante
Disclaimer: Illustrations in Watercolor are painted by Eleonora Escalante. Other types of illustrations or videos (which are not mine) are used for educational purposes ONLY. Nevertheless, the majority of the pictures, images, or videos shown on this blog are not mine. I do not own any of the lovely photos or images posted unless otherwise stated.